
Scientists have successfully created methanetetrol, a highly unstable molecule known as a “super alcohol,” in a lab for the first time. Previously only theorized and possibly existing in space, this compound has four hydroxyl (OH) groups bonded to one carbon—an extremely rare structure. By mimicking deep space conditions, researchers managed to synthesize it, offering insights into the kinds of molecules that could kickstart prebiotic chemistry and potentially lead to life in the universe.

What’s the story behind this “super alcohol”?
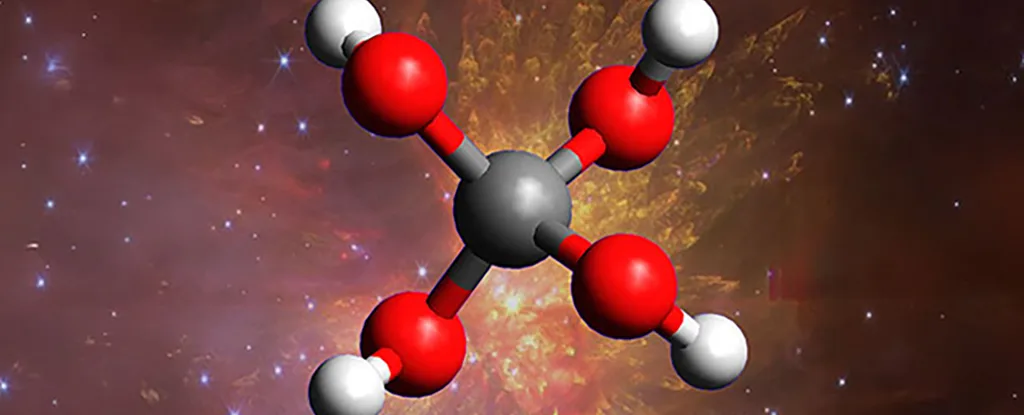
Scientists have successfully synthesized methanetetrol (chemical formula C(OH)₄), a molecule long theorized but never before observed—especially not on Earth.
- Methanetetrol is an ortho acid, featuring four hydroxyl (OH) groups attached to a single carbon atom—an extremely unstable configuration that resists bonding, making it notoriously difficult to isolate.
- Researchers from the University of Hawaiʻi at Mānoa (led by Ralf Kaiser), alongside collaborators from the University of Mississippi and Florida International University, recreated the harsh conditions of deep space in the lab to create this compound. They froze water and carbon dioxide into ice near absolute zero and bombarded it with radiation simulating cosmic rays.
- The resulting molecule—released into gas form—was detected using powerful ultraviolet spectroscopy techniques.
Why does methanetetrol matter?
- Astrochemical breakthrough: This marks the first laboratory synthesis of such a complex and previously elusive molecule, expanding our understanding of what chemistry is possible in the frigid, vacuum-like conditions of space.
- A “seed of life”: Described by researchers as a “prebiotic concentrate” or an “acorn that could grow into a tree,” methanetetrol may represent a starting point for more complex chemical processes that could lead toward life.
- Instability with potential: Its inherent instability means that when energized, methanetetrol could decompose into key molecules like water and hydrogen peroxide—both vital for prebiotic chemistry.
- Guide for space discovery: Now that we know it can exist, astronomers may start looking for methanetetrol in interstellar environments—such as icy dust clouds between stars—offering new clues in the search for life-supporting chemistry.

In summary
Scientists have recreated for the first time on Earth a rare and unstable molecule—methanetetrol—under conditions mimicking space. This “super alcohol” isn’t just a chemical oddity; it’s a potential keystone in understanding how complex organic chemistry—and possibly life—can emerge in the cosmos.



